In silico ADMET Profiling of Flavonoids as Potential Phosphodiesterase-5 Inhibitors in the Management of Erectile Dysfunction
DOI:
https://doi.org/10.26538/tjdr/v3i1.1Keywords:
Flavonoids, Adsorption, Distribution, Toxicity, In silico, Apigenin, Kaempferol, NaringinAbstract
Purpose: Erectile dysfunction (ED) affects millions of men globally, and its pharmacological management relies mainly on agents such as sildenafil, tadalafil, avanafil, and vardenafil. Although effective, these drugs are associated with adverse effects, creating a need for therapeutic alternatives with improved safety, efficacy, and bioavailability profiles. This study evaluated the preclinical absorption, distribution, metabolism, excretion, and toxicity (ADMET) profiles of the flavonoids apigenin, kaempferol, and naringin as potential oral therapeutic candidates for ED.
Methods: In silico ADMET profiling was conducted using SwissADME and ProTox-II to predict key pharmacokinetic and safety parameters, including solubility, gastrointestinal absorption, blood–brain barrier permeability, cytochrome P450 interactions, drug-likeness, and toxicity risks. Sildenafil served as the reference standard.
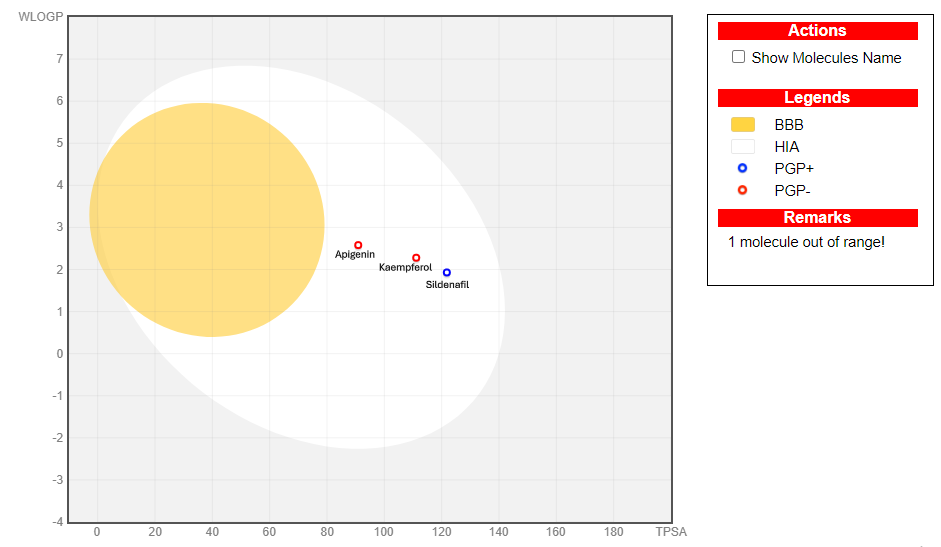
Results: Apigenin and kaempferol exhibited favorable oral bioavailability and drug-likeness, consistent with Lipinski’s, Veber’s, Egan’s, and Muegge’s rules. These properties were supported by their optimal molecular weights, moderate topological polar surface areas, and minimal molecular flexibility (apigenin: 270.24 g/mol, TPSA 90.90 Å2; kaempferol: 286.24 g/mol, TPSA 111.13 Å2). Naringin, however, displayed limited absorption potential due to its higher molecular weight (580.53 g/mol) and large polar surface area (225.06 Å2). All compounds demonstrated acceptable predicted safety profiles, with no signals for mutagenicity or hepatotoxicity.
Conclusion: The findings indicate that apigenin, kaempferol, and naringin may offer safer, more accessible alternatives or adjuncts to conventional ED medications such as sildenafil. Their favorable predicted pharmacokinetic and toxicity profiles support further exploration, and experimental validation in in vitro and in vivo models is recommended to confirm therapeutic potential.
Downloads
References
1. Mazzilli F. Erectile dysfunction: causes, diagnosis and treatment: an update. J Clin Med. 2022; 11(21):6429. https://doi.org/10.3390/jcm11216429
2. Argiolas A, Argiolas FM, Argiolas G, Melis MR. Erectile dysfunction: treatments, advances and new therapeutic strategies. Brain Sci. 2023; 13(5):802. https://doi.org/10.3390/brainsci13050802
3. Kaltsas A, Dimitriadis F, Zachariou A, Sofikitis N, Chrisofos M. Phosphodiesterase type 5 inhibitors in male reproduction: molecular mechanisms and clinical implications for fertility management. Cells. 2025; 14(2):120. https://doi.org/10.3390/cells14020120
4. Özkent MS, Hamarat MB, Taşkapu HH, Kılınç MT, Göger YE, Sönmez MG. Is erectile dysfunction related to self‐esteem and depression? A prospective case–control study. Andrologia. 2021; 53(1):e13910.
5. De Nunzio C, Roehrborn CG, Andersson KE, McVary KT. Erectile dysfunction and lower urinary tract symptoms. Eur Urol Focus. 2017; 3(4-5):352-63. https://doi.org/10.1016/j.euf.2017.11.004
6. Alrumaihi F, Raut R, Yahia EA, Kumar V, Anwar S. A review on risk factors, diagnostic innovations, and plant based therapies for the management of erectile dysfunction. Uro. 2024; 4(2):60-88. https://doi.org/10.3390/uro4020006
7. Allen MS, Tostes RC. Cigarette smoking and erectile dysfunction: an updated review with a focus on pathophysiology, e-cigarettes, and smoking cessation. Sex Med Rev. 2023; 11(1):61-73. https://doi.org/10.1093/sxmrev/qeac007
8. Saikia Q, Hazarika A, Mishra R. A review on the pharmacological importance of PDE5 and its inhibition to manage biomedical conditions. J Pharmacol Pharmacother. 2022; 13(3):246-57. https://doi.org/10.1177/0976500X221129008
9. Paronetto MP, Crescioli C. Rethinking of phosphodiesterase 5 inhibition: the old, the new and the perspective in human health. Front Endocrinol. 2024; 15:1461642. https://doi.org/10.3389/fendo.2024.1461642
10. Sin VJ, Anand GS, Koh HL. Botanical medicine and natural products used for erectile dysfunction. Sex Med Rev. 2021; 9(4):568-92.
11. Singh A, Singh J, Parween G, Khator R, Monga V. A comprehensive review of apigenin a dietary flavonoid: biological sources, nutraceutical prospects, chemistry and pharmacological insights and health benefits. Crit Rev Food Sci Nutr. 2025; 65(23):4529-65.
https://doi.org/10.1080/10408398.2024.2390550
12. Chen J, Zhong H, Huang Z, Chen X, You J, Zou T. A critical review of kaempferol in intestinal health and diseases. Antioxidants. 2023; 12(8):1642. https://www.mdpi.com/2076-3921/12/8/1642
13. Salehi B, Fokou PV, Sharifi-Rad M, Zucca P, Pezzani R, Martins N, Sharifi-Rad J. The therapeutic potential of naringenin: a review of clinical trials. Pharmaceuticals. 2019; 12(1):11. https://doi.org/10.3390/ph12010011
14. Venkateswara Rao P, Kiran SD, Rohini P, Bhagyasree P. Flavonoid: A review on Naringenin. J. Pharmacogn. Phytochem. 2017; 6:2778-83.
15. Bayram P, Aksak Karamese S, Ozdemir B, Salum C, Erol HS, Karamese M. Two flavonoids, baicalein and naringin, are effective as anti-inflammatory and anti-oxidant agents in a rat model of polymicrobial sepsis. Immunopharmacol Immunotoxicol. 2023; 45(5):597-606. https://doi.org/10.1080/08923973.2023.2197143
16. Thomas SD, Jha NK, Jha SK, Sadek B, Ojha S. Pharmacological and molecular insight on the cardioprotective role of apigenin. Nutrients. 2023; 15(2):385. https://doi.org/10.3390/nu15020385
17. Zhuang J, Wang J, Zhang B, Chai D, Zuo Z. The prophylactic effects of naringin on steroid-induced early-stage osteonecrosis in rats: a preliminary study. Cell Mol Biol. 2023; 69(5):94-104. http://dx.doi.org/10.14715/cmb/2023.69.5.16
18. Stabrauskiene J, Kopustinskiene DM, Lazauskas R, Bernatoniene J. Naringin and naringenin: Their mechanisms of action and the potential anticancer activities. Biomedicines. 2022; 10(7):1686. https://doi.org/10.3390/biomedicines10071686
19. Imran M, Salehi B, Sharifi-Rad J, Aslam Gondal T, Saeed F, Imran A, Shahbaz M, Tsouh Fokou PV, Umair Arshad M, Khan H, Guerreiro SG. Kaempferol: A key emphasis to its anticancer potential. Molecules. 2019; 24(12):2277. https://doi.org/10.3390/molecules24122277
20. Reddy KA, Ashma M, Jyothi V, Jyostna TS. Molecular Properties Prediction of Phenothiazine Derivatives by Using Swiss ADME, PkCSM, Lazar and Protox. World J Pharm Sci. 2019; 7(11):65-71. [cited 2025 Oct. 1]. Available from: https://wjpsonline.com/index.php/wjps/article/view/molecular-properties-prediction-phenothiazine-derivatives
21. Riyadi PH, Sari ID, Kurniasih RA, Agustini TW, Swastawati F, Herawati VE, Tanod WA. SwissADME predictions of pharmacokinetics and drug-likeness properties of small molecules present in Spirulina platensis. IOP conference series: earth and environmental science 2021; 890(1):012021. IOP Publishing. https://doi.org/10.1088/1755-1315/890/1/012021
22. Yusuf SE. Pharmacokinetic Evaluation of Sulfadicramide through SwissADME: A Computational Insight into Drug-Likeness and Bioavailability. MAS J Appl Sci. 2025; 10(2):357-62. http://dx.doi.org/10.5281/zenodo.15741944
23. Mahanthesh M, Ranjith D, Sreedhara J, Kumar Wodeyar DS, Hebbar A, Sudharani R, Ramesh BK, Mahesh C, Doddamani PL, Ram J, Radha J. In Silico assessment of Tolfenpyrad utilizing Swiss-ADME, Pass and Molinspiration: A comprehensive analysis. Int J Vet Sci Anim Husb. 2024; 9(4):653-9.
24. Banerjee P, Eckert AO, Schrey AK, Preissner R. ProTox-II: a webserver for the prediction of toxicity of chemicals. Nucleic Acids Res. 2018; 46(W1):W257-63. https://doi.org/10.1093/nar/gky318
25. Suliman RS, Alghamdi SS, Ali R, Rahman I, Alqahtani T, Frah IK, Aljatli DA, Huwaizi S, Algheribe S, Alehaideb Z, Islam I. Distinct mechanisms of cytotoxicity in novel nitrogenous heterocycles: future directions for a new anti-cancer agent. Molecules. 2022; 27(8):2409. https://doi.org/10.3390/molecules27082409
26. Daina A, Michielin O, Zoete V. SwissADME: a free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci Rep. 2017; 7(1):42717. https://doi.org/10.1038/srep42717
27. İslamoğlu F, Hacıfazlıoğlu E. Investigation of the usability of some triazole derivative compounds as drug active ingredients by ADME and molecular docking properties. Moroc J Chem. 2022; 10(4):J-Chem. https://doi.org/10.48317/IMIST.PRSM/morjchem-v10i3.30855
28. Nabati M, Bodaghi-Namileh V. Design of novel drugs (P3TZ, H2P3TZ, M2P3TZ, H4P3TZ and M4P3TZ) based on zonisamide for autism treatment by binding to potassium voltage-gated channel subfamily D member 2 (Kv4. 2). Int J New Chem. 2019; 6(4):254-76.
29. Delaney JS. ESOL: estimating aqueous solubility directly from molecular structure. J Chem Inf Comput Sci. 2004; 44(3):1000-5. https://doi.org/10.1021/ci034243x
30. Ali J, Camilleri P, Brown MB, Hutt AJ, Kirton SB. Revisiting the general solubility equation: in silico prediction of aqueous solubility incorporating the effect of topographical polar surface area. J Chem Inf Model. 2012; 52(2):420-8. https://doi.org/10.1021/ci200387c
31. Oyeneyin, Oluwatoba, Nureni Ipinloju, Nathanael Ojo, and Daniel Akerele. "Structural modification of ibuprofen as new NSAIDs via DFT, molecular docking and pharmacokinetics studies." Int J Adv Eng Pure Sci. 2021; 33(4): 614-626. https://doi.org/10.7240/jeps.928422
32. Bhuia MS, Eity TA, Chowdhury R, Ansari SA, Bappi MH, Nayeem MA, Akter F, Islam MT. Anxiolytic Activity of Morellic Acid: Modulation of Diazepam's Anxiolytic Effects, Possibly Through GABAergic Interventions. CNS Neurosci Ther. 2025;
31(2):e70276. https://doi.org/10.1111/cns.70276
33. Brenk R, Schipani A, James D, Krasowski A, Gilbert IH, Frearson J, Wyatt PG. Lessons learnt from assembling screening libraries for drug discovery for neglected diseases. ChemMedChem: Chem Enabl Drug Discov. 2008; 3(3):435-44. https://doi.org/10.1002/cmdc.200700139
34. Kralj S, Jukič M, Bren U. Molecular filters in medicinal chemistry. Encyclopedia. 2023; 3(2):501-11. https://doi.org/10.3390/encyclopedia3020035
35. Banerjee P, Kemmler E, Dunkel M, Preissner R. ProTox 3.0: a webserver for the prediction of toxicity of chemicals. Nucleic Acids Res. 2024; 52(W1):W513-20. https://doi.org/10.1093/nar/gkae303
36. Tahir A, Saleh YI, Muazu IK. Structure-Based Repurposing of Clinically Approved Compounds for PDE-5 Inhibition: Integrative Computational and Pharmacokinetic Evaluation. Al-Mustaqbal J Pharm Med Sci. 2025; 3(3):5. https://doi.org/10.62846/3006-5909.1036
37. Stielow M, Witczyńska A, Kubryń N, Fijałkowski Ł, Nowaczyk J, Nowaczyk A. The Bioavailability of Drugs—The Current State of Knowledge. Molecules. 2023; 28(24):8038. https://doi.org/10.3390/molecules28248038
Downloads
Published
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.









